June was Alzheimer’s Awareness month and people all over were wearing purple to show their support for those suffering from the terrible disease. Now, it looks like a miracle drug might be just around the corner.
Tony Bennett and Wife Susan Benedetto Wear Purple for Alzheimer's and Brain Awareness Month https://t.co/eIMxa6SVZe
— People (@people) June 2, 2022
The FDA wrote, “Leqembi is the second of a new category of medications approved for Alzheimer’s disease that target the fundamental pathophysiology of the disease. These medications represent an important advancement in the ongoing fight to effectively treat Alzheimer’s disease.
“Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones,” said Billy Dunn, M.D., director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research. “This treatment option is the latest therapy to target and affect the underlying disease process of Alzheimer’s, instead of only treating the symptoms of the disease.”
Leqembi was approved using the Accelerated Approval pathway, under which the FDA may approve drugs for serious conditions where there is an unmet medical need and a drug is shown to have an effect on a surrogate endpoint that is reasonably likely to predict a clinical benefit to patients. The results of a Phase 3 randomized, controlled clinical trial to confirm the drug’s clinical benefit have recently been reported and the agency anticipates receiving the data soon.”
CNN explained the process. “Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones,” Dr. Billy Dunn, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, said in a statement. “This treatment option is the latest therapy to target and affect the underlying disease process of Alzheimer’s, instead of only treating the symptoms of the disease.”
Lecanemab will be marketed as Leqembi, the FDA statement said. It has shown “potential” as an Alzheimer’s disease treatment by appearing to slow progression, according to Phase 3 trial results, but it has raised safety concerns due to its association with certain serious adverse events, including brain swelling and bleeding.
In July, the FDA accepted Eisai’s Biologics License Application for lecanemab under the accelerated approval pathway and granted the drug priority review, according to the company. The accelerated approval program allows for earlier approval of medications that treat serious conditions and “fill an unmet medical need” while the drugs continue to be studied in larger and longer trials.
If those trials confirm that the drug provides a clinical benefit, the FDA could grant traditional approval. But if the confirmatory trial does not show benefit, the FDA has the regulatory procedures that could lead to taking the drug off the market.
The new drug is not without risks, however. The New York Times said, “Concerns about safety have been stoked by news reports of the deaths of three patients who experienced brain swelling and brain bleeding, two of whom were being treated with blood thinners. Those patients participated in a large Phase 3 trial of the drug, during which they were not told whether they received it or a placebo. But their deaths occurred after that phase of the trial, when they were knowingly being treated with lecanemab in what’s known as an open-label extension study.
One case, the subject of a report this week in the New England Journal of Medicine, involved a 65-year-old woman who had a stroke and, after receiving a standard treatment for stroke-related blood clots known as t-PA, experienced serious brain bleeding and died a few days later. In an earlier article about the case in the journal Science, a neuropathologist who conducted an autopsy said he believed that Leqembi weakened her blood vessels and made them vulnerable to bursting when she received the blood clotting treatment.
In a published letter responding to the New England Journal of Medicine report, two researchers involved in Eisai’s Leqembi trial asserted that “t-PA appears to be the proximate cause of death,” not Leqembi, and noted that the woman had two copies of a gene mutation that increases brain swelling risk with anti-amyloid treatments. But, they said, we agree that this case raises important management issues for patients with Alzheimer’s disease.’”
“The clinical trial results, published in the New England Journal of Medicine, found that cognitive decline was 27% slower over 18 months in people who received lecanemab compared with those who did not receive the treatment. The study was funded by Biogen and Eisai,” according to CNBC.
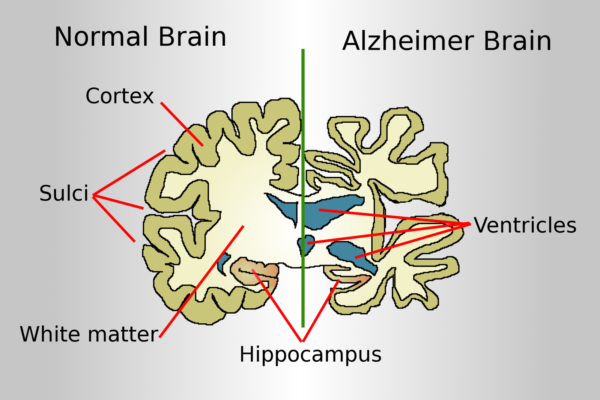
Over 6.5 million people in the United States suffer from Alzheimer’s. Worldwide almost 50 million people have some stage of the illness. Any slowing of the disease can at least be celebrated as progress towards a final cure.









Mass produce RX ASAP